 
The tubes were centrifuged at 600 g for 3 min at 4 ☌ to pellet nuclei and cell debris. Each homogenate was split into two 55 μL aliquots and transferred into two fresh Protein LoBind tubes. Cell and organelle lysis was completed by incubation for 10 min on ice. The cells were pelleted with 300 g for 10 min at 4 ☌ and resuspended by pipetting 15× up and down in 120 μL high-density lysis buffer (25 mM HEPES-KOH pH 7.2, 110 mM potassium acetate, 5 mM magnesium acetate, 1 mM EGTA, 25% sucrose, 5% glycerol, 1 mM 1,4-dithiothreitol, 1× cOmplete EDTA-free protease inhibitor cocktail, 0.1 mg/mL cycloheximide, 0.015% digitonin, and 400 U/mL RiboLock RNase inhibitor ). HEK 293T cells (1 million), mouse hybridoma cells (1 million of 5E4, KT13, and KT22 cells mixed in ratio 1:1:1), ARH-77 leukemia cells (ATCC CRL-1621, 1 million), or freshly isolated human CD19 + B cells from pre- and post Td-booster immunization samples (1.5 million each) were resuspended in 1 mL PBS with 50 μg/mL cycloheximide and incubated for 10 min to stall ribosomes with associated messenger RNAs (mRNA) at the rough endoplasmic reticulum. Protein LoBind 1.5 mL centrifuge tubes (Eppendorf) were used to minimize cell adhesion to the tube walls. Cycloheximide treatment and microsome preparationĪll pipetting steps were performed on ice and centrifugations were carried out at 4 ☌ using an Eppendorf 5810R centrifuge with fixed angle rotor F-45-30-11. Antibody HC and LC sequences from individual hybridomas were determined by reverse transcription polymerase chain reaction (RT-PCR) and capillary sequencing (Eurofins Genomics). HEK 293T and hybridoma cells were grown in DMEM (Gibco) supplemented with 13% FBS (Gibco), 1× Penicillin/Streptomycin (Thermo Fisher), and 1× GlutaMAX (Gibco). Mouse hybridoma cell line 5E4/1F1 was kindly provided by Miha Kosmač and Vladka Čurin Šerbec (University of Ljubljana). Both cell lines were deposited to the DSHB by Kazumasa Takeda and Asako Sugimoto (DSHB hybridoma products KT13 and KT22). Mouse hybridoma cell lines KT13 and KT22 were obtained from the Developmental Studies Hybridoma Bank (DSHB). The HEK 293T cell line was obtained from the American Type Culture Collection (ATCC CRL-3216). This method makes paired Ig sequencing widely applicable even for laboratories without specialized equipment and personnel. Here, we describe a high-throughput method which enables sequencing of paired HC-LC immunoglobulin (Ig) repertoires from millions of B cells simply by using a cooled table-top centrifuge, a magnetic stirrer, and a thermal cycler. Although substantial, the existing methods are limited by their commercial availability, high costs, and require an elaborate construction of flow-focusing or microfluidic devices and dedicated personnel for operation. More recently, two emulsion-based methods reported paired HC-LC repertoire sequencing from 2–3 × 10 6 B cells at single-cell level. 
To this end, several single-cell paired sequencing technologies were reported more recently, which were initially limited by low cell numbers (< 400–10 5 cells) and sometimes required the use of complex microfluidic systems however, cellular throughput is improving through newer developments, such as droplet-based systems and the 10× Genomics platform. 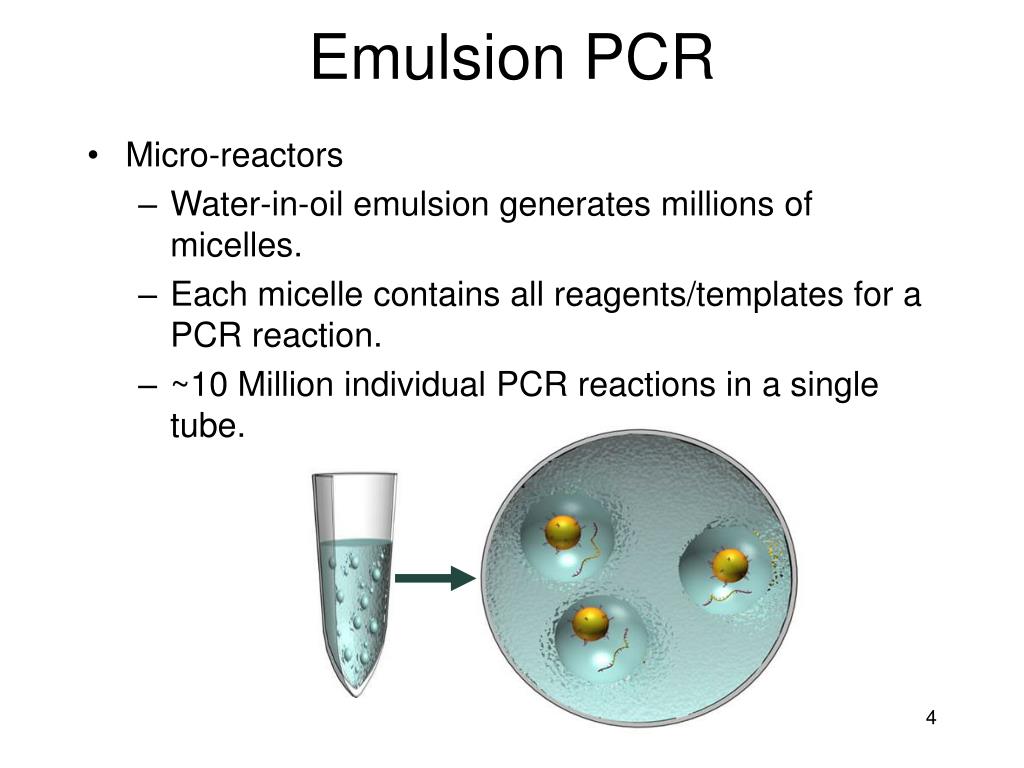
Retaining paired HC-LC data from bulk B cell populations at single-cell level remained a major obstacle for a long time. Initially, antibody repertoire analysis focused on obtaining information from antibody heavy chains (HC) only, missing the native light-chain (LC) pairing information that is necessary for antibody cloning and expression. High-throughput sequencing of immunoglobulin repertoires from B cells has emerged as a powerful tool to investigate repertoire changes for antibody discovery, vaccine efficacy studies, and in other healthcare applications. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
